Monitoring Report Template Clinical Trials. It is crucial that investigators seek the guidance of with their native institutional review board for any institution-specific templates and/or necessities pertaining to the format and content of the consent document. It is estimated that kind 1 diabetics expertise two delicate, symptomatic episodes of hypoglycemia per week. Activities include evaluating the affected person inhabitants pool, catchment space description, recruitment plan, and target enrollment for any new scientific websites. The monitoring report template ought to be open throughout the go to, each to keep you on track and to provide you a spot to write notes.
A clinical trial document tracking log can help you keep a written trail of the documents and after they have been submitted and approved. If a affected person is aware and in a place to swallow safely, food or drink may be administered, as properly as glucose tabs or gel. The procedures to protect in opposition to risks include (e.g., a secure, hygienic environment for all medical procedures and an skilled, certified staff).
An important element of making ready is to have a look at previous scientific monitoring stories and audit reviews, if any have been performed. Hypoglycemia is widespread in folks with sort 1 diabetes, and in people with type 2 diabetes taking insulin, glinides, or sulfonylurea. I noted just a single need of this methodology – cause offense intricacy of the code… This is the entire purpose for doing the positioning monitoring, so take notes that reveal adherence to protocol, good clinical practices , and rules. Also helpful to establish if the topic consented for the pattern to be retained for future research.
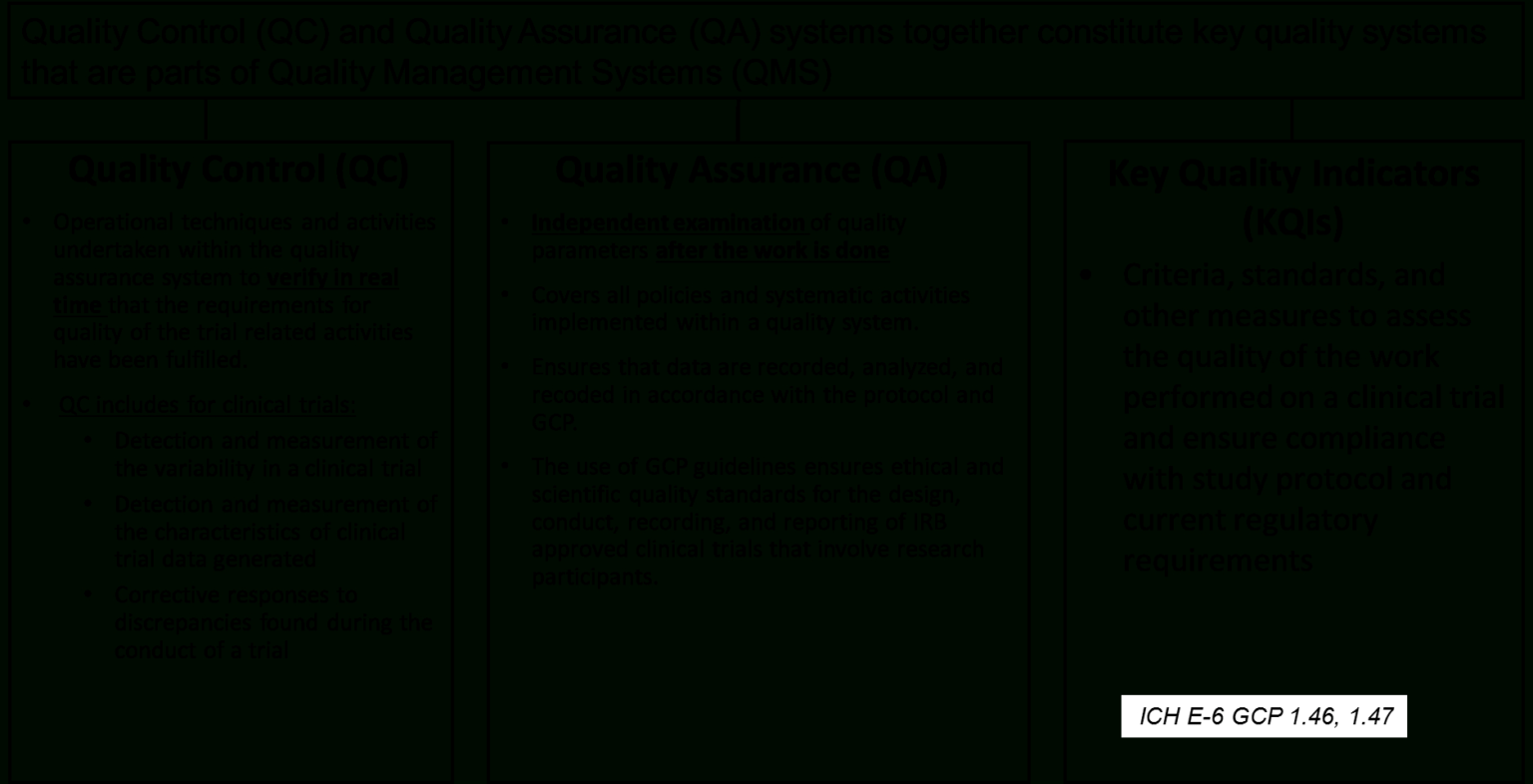
Provides directions for constant examine procedure implementation and knowledge assortment across members and scientific sites. Details the study’s group, operations, research procedures, data management, and high quality management. Clinicalstudies use a collection of case report forms to gather knowledge in a consistent manner.

Much appreciated, and I hope you discovered every thing you wanted in your SOP on research misconduct. These templates and tools are ordered by category, so please scroll down to find what you need. SharePoint Server allows the group to manage the SharePoint features obtainable to employees, and you can scale it to fulfill different numbers of users.
Good Scientific Apply Coaching
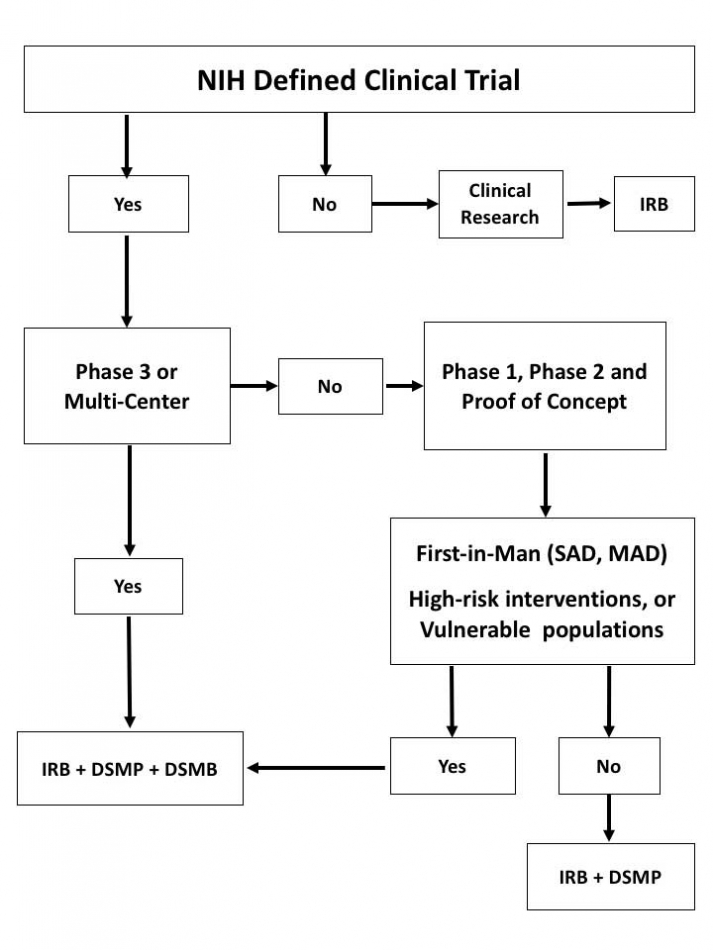
As part of the NIAID system for overseeing and monitoring clinical trials, NIAID and the awardee jointly determine the monitoring kind earlier than a scientific trial begins. A few purposes require the putting away of Waste Management Report Template in a database.

Research Highlights Stories on research being performed by researchers at NIMH-supported institutions across the nation and researchers in the NIMH Division of Intramural Research Programs . Science News List of NIMH science information together with press releases, science updates and institute bulletins. Priority Research Areas Learn about NIMH precedence areas for research and funding which have the potential to improve psychological health care over the short, medium, and long term.
Templates For Developing A Guide Of Working Procedures Moop
Writing efficient monitoring reports requires an in-depth data of the job, the research protocol, the positioning, their practices, the sponsor’s procedures, the monitoring plan, the report template and, in fact, the regulations. The Toolbox incorporates templates, sample forms, and knowledge materials to assist medical investigators in the development and conduct of high-quality medical research studies. Upon the off likelihood that you’re not using templates after that you are sitting relating to idly.

If UPs occur through the study, they will be reported to the IRB, Monitoring Body, and NIAMS by the study group. The UP report for the Monitoring Body and NIAMS shall be accomplished and submitted to the NIAMS Executive Secretary within 48 hours of the investigator turning into conscious of the event.
Those with hypoglycemic unawareness experience the symptoms of hypoglycemia at far lower ranges of blood glucose. The hypoglycemic particular person not solely features consciousness of hypoglycemia at very low blood glucose levels, however additionally they require excessive ranges of carbohydrates or glucagon to recover their blood glucose to normal levels.

Meetings and Events Details about upcoming events — including conferences, conferences, workshops, lectures, webinars, and chats — sponsored by the NIMH. Clinical Research Find the latest NIH and NIMH insurance policies, steerage, and assets for medical analysis. Research Conducted at NIMH The Division of Intramural Research Programs is the interior research division of the NIMH.
Gallery Of Monitoring Report Template Clinical Trials Superior 10 Medical Research Coordinator Resume Example Collection Resume
This document provides a log template for monitoring subject-specific protocol deviations/violations. If captured electronically, subject-specific deviation logs may be exported into a study-wide deviation log. Monitoring Report Template Clinical Trials

A few functions require the putting away of Cleaning Report Template in a database. This rearranges template maintain – all reviews are put away in one spot, and permits to remove the doorway rights to varied templates.

The goal of progress stories would be to build up and outline upgrades, key sides, together with summaries of a unbroken trial. It’s very crucial to bear in mind that progress reviews have to be filed to institutional evaluation board/independent integrity questionnaire (IRB/IEC), following a trial that has obtained positive opinion. Such a report ought to be rigorously ready and it should summarize the means that a examine is completed.
PDF Generator accompanies fundamental instigation and easy to make the most of interface. It makes stories in PDF viewpoint legitimately from MSSQL or MySQL databases, csv, txt chronicles or from bodily entered info. It is conceivable taking into account an application called PDF Generator.
Perhaps your realized already has a license in imitation of a clipart company that permits them to use clipart on moot documents. Check as soon as the moot secretary to look if that is something you must use in your template .
- Resources for Researchers Information about resources similar to information, tissue, model organisms and imaging sources to help the NIMH analysis neighborhood.
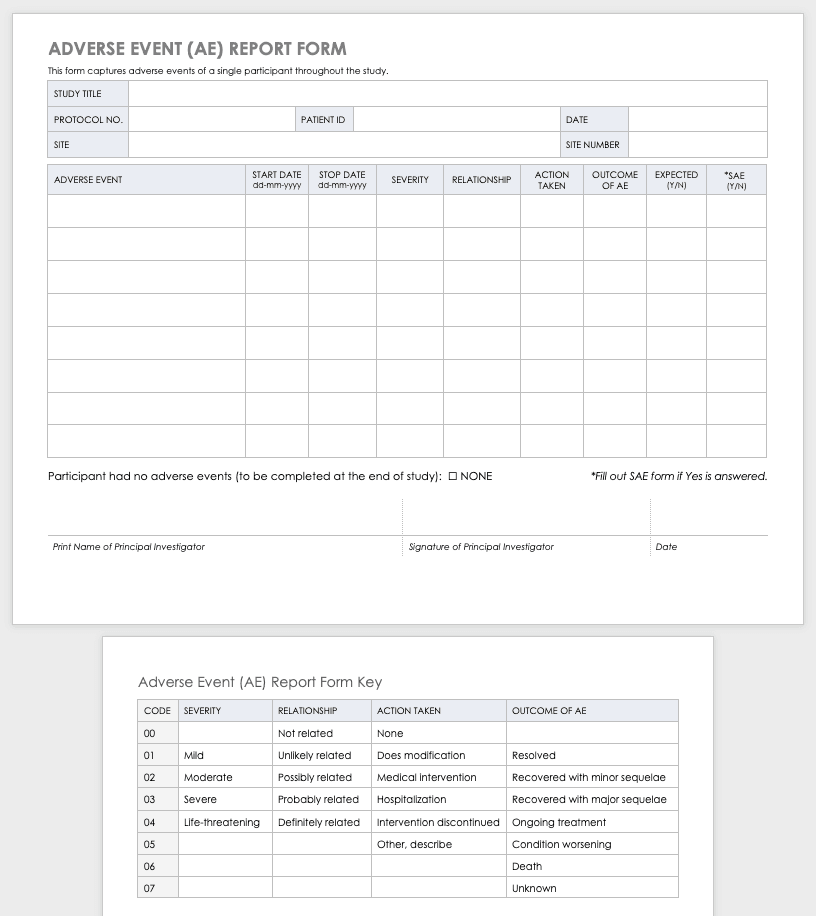
- Other relevant medical data shall be completed on all SAEs regardless of expectedness or relatedness.
- It makes stories in PDF slant legitimately from MSSQL or MySQL databases, csv, txt historical past or from bodily entered data.
- Data and security monitoring reports are despatched to the Monitoring Body and the NIAMS prematurely of the semi-annual conferences and can embrace a detailed evaluation of study progress, information and safety issues.
- The interim look is really helpful on the end of yr one as we anticipate approximately 50% of the participants followed for no much less than six months.
- Science Education Use these free digital, outreach supplies in your neighborhood and on social media to unfold the word about psychological well being.
Several essential documents should be organized and retained for clinical studies. These organized paperwork are referred to as the Regulatory Binder , which is retained at the Investigator’s scientific web site.
It is conceivable to utilize pre-characterized Monitoring Report Template Clinical Trials and any sort of steering source to create reviews in a quick timeframe. For steering on NIAID scientific trial monitoring requirements, see NIAID Clinical Terms of Awardand follow all application instructions. Guidelines and templates for developing a scientific study MOOP to facilitate consistency in protocol implementation and information assortment throughout employees, sufferers and clinical websites.

All the extra considerably, it furnishes companions and speculators much like a diagram of what’s happening in a business. This will rule their pledge to your situation for the considering yr and whether or not they look potential for growth inside the group. To show your thoughts obviously and in an intelligent method, you want to type out a yearly Monitoring Report Template Clinical Trials for those which would possibly be keen on auditing your business.

This part supplies information on how dangers to members might be managed. It should specify any events that might preclude a participant from continuing in the examine.

You will want to get this to make definite that something you place upon the template will print out okay. Taking into consideration you might have previewed how it will print, you can both create some changes (reducing font measurement, lowering clipart size, and so forth.) or simply go forward and print out Monitoring Report Template Clinical Trials.
The presence of instructional calisthenics permits the people who have by no means utilized any comparable programming to make reviews to begin making stories the least demanding and quickest method. The presence of pre-characterized template allows you to see the lively all right of the appliance and that is the fastest technique to perceive how the reports would resemble.

Aside from the ethics committee, researchers may also must present yearly enchancment stories of an investigation to spouses, encouraging associations, and/or organizations, together with other interested events if wanted.

In this article, you will discover everything you have to begin your medical analysis trials, with easy-to-understand guidance and terminology, 26 adaptable templates, and project plans in Microsoft Word, Excel, Project, and SharePoint formats. Hypoglycemia was first found by James Collip when he was working with Frederick Banting on purifying insulin in 1922.

Sample kind to record any unanticipated problems that occur during the study. Provides an at-a-glance reference for including important documents for scientific research in a Regulatory Binder.

These greatest practices apply to all clinical analysis, whether or not the research falls beneath IND rules or different regulatory regimes. Provides directions for organizing essential documents for medical research in a Regulatory Binder.

This is the entire reason for doing the positioning monitoring, so take notes that show adherence to protocol, good scientific practices , and laws. Clinical research may find yourself in problems for the members and set off an adverse or severe adverse event.

It should embrace an outline of all expected opposed occasions , the recognized unwanted facet effects of the intervention, and all identified risks or issues of the outcomes being assessed. This document provides a common outline of a study-specific knowledgeable consent doc. It is essential that investigators seek the guidance of with their local institutional review board for any institution-specific templates and/or requirements pertaining to the format and content material of the consent doc.
Sometimes yellows or purples can arrive out light, although they see welcome upon your laptop monitor. Review upfront of the examine initiation the research particular stopping guidelines and plans for interim analyses as established by the PI and selected members of the study team. These plans outline the conditions under which a research may be stopped (e.g., difficulties in recruitment, retention, obtaining end result measures, or different issues).
Template are agreed the spine to students learning and greedy ideas taught by the instructor. Making your own template is straightforward, and it lets you mix simply the best material that you simply want…
Instead, the kidneys are answerable for removing insulin from the physique, and when this operate is impaired in kidney failure, the insulin stays in circulation longer, leading to hypoglycemia. This ensures that these performing duties have a clear plan of what, when and howtrial activities are undertaken and likewise allows auditors/inspectors to traditionally reconstruct all trial management activities. The NIMH Data Archive is an informatics platform for the sharing of de-identified human subject information from all scientific analysis funded by the NIMH.

These templates help the principal investigator in the design of their knowledgeable consent types . You can adapt them to accommodate the small print of any study and include each the knowledge sheet and the consent form.
First launched in 1985, Project is a well-respected Microsoft product for project administration. Microsoft Project was not historically available as a half of Office Suites, a package of applications for professionals and skilled organizations.
Collip noticed that if he injected rabbits with a too massive a dose of insulin, the rabbits started convulsing, went right into a coma, after which died. He defined one unit of insulin as the amount necessary to induce this convulsing hypoglycemic response in a rabbit.



